snorestops
SnoreStop® Anti-Snoring Chewable Tablets
Target Snoring Naturally without Devices or Discomfort
Description
Description




Quiet Nights. Natural Relief. Since 1995.
Snoring doesn’t just break up your sleep it can break up your peace of mind. Elbow nudges, exhausted mornings, and even separate bedrooms are often caused by one simple thing: blocked airways.
SnoreStop Chewable Tablets are the natural, device-free solution that targets snoring at the source. With just two tablets at bedtime, you can reduce inflammation to help open airways and dry out built up mucus to enjoy the kind of deep, restorative sleep you (and your partner) deserve.
Why People Love SnoreStop Tablets
-
Targets Real Source of Snoring – Helps reduce inflammation in the airway(s) and helps dry out built up mucus so you can breathe freely and sleep quietly.
-
Natural & Safe – Plant-based formula, safe for nightly use.
-
Fast & Effortless – Chew two tablets before bed. No water. No hassle.
-
Discreet & Travel-Friendly – Take anywhere for quiet nights without bulky devices.
-
Trusted & Proven – Clinically tested and relied on by couples since 1995.
Unlike mouth guards, chin straps, or taping your mouth shut, SnoreStop works where the problem really starts: inside your airways. No devices. No discomfort. Just quiet, natural relief.
Imagine waking up to:
✔ Quiet, uninterrupted nights
✔ Energy and focus that last all day
✔ No more partner frustration or sleeping apart
✔ The confidence of a solution that’s safe, natural, and proven
SnoreStop Chewable Tablets — effortless, natural, and trusted for 30 years.
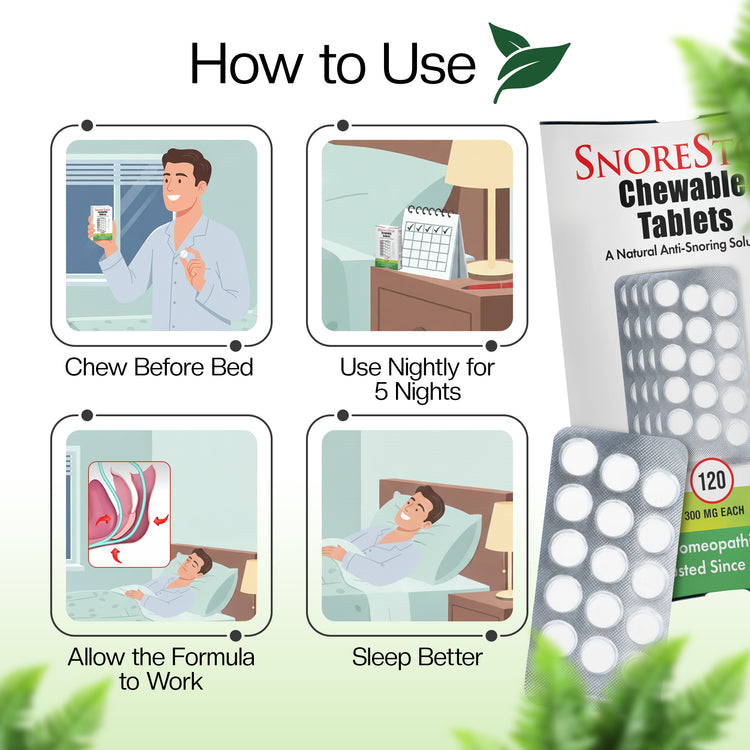
Usage Instructions
Directions for Use:
- Chew one tablet 30 minutes before bedtime and repeat at bedtime.
- Do not take with water; chew or suck tablets.
- 5-7 day treatment course for optimal results.
Treatment Timeline:
- 5-7 Day Course: While some experience results immediately, most users notice improvements within a minimum of 5 nights of consistent use.
Pro Tips for Best Results
- Take at bedtime, 30 minutes apart from food, water, or medicine.
- Stay hydrated during the day for comfort at night.
- Avoid sedatives, alcohol, or drowsy medication at bedtime.
Experience better breathing and restful nights within a week of using SnoreStop.
* Individual results may vary. SnoreStop does not treat sleep apnea. Results can depend on a variety of factors including overall health, diet, and other lifestyle factors.
Warnings: Use only as directed. Keep out of reach of children. Do not use it on children under 12 years of age. If pregnant or breastfeeding, ask a doctor before using it. Stop using it and ask a doctor if symptoms do not improve in 7 days.
Ingredients
Ingredients
Ingredients & Purpose:
- Nux Vomica 4X HPUS: helps open constricted pharynx
- Belladonna 6X HPUS: helps decongest enlarged tonsils
- Ephedra vulgaris* 6X HPUS: helps relieve congestion
- Hydrastis Canadensis 6X HPUS: helps decongest swollen tongue
- Kali Bichromicum 6X HPUS: helps relieve stuffy nose
- Teucrium marum 6X HPUS: helps open air passages
- Histaminum hydrochloricum 12X HPUS: natural antihistamine
Dilution Scale: 6X is equal to one part per million. * Relax knowing that SnoreStop contains no ephedrine, pseudoephedrine, or alkaloids. Review our Certificate of Analysis for confirmation. Your peace of mind is paramount to us. With a proven safety record since 1995, SnoreStop is your trusted choice. The letters “HPUS” indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Chewable Tablets Certificate of Analysis
Inactive ingredients: Anhydrous Lactose (3mg/tab), Croscamellose sodium, Dextrose (45 mg/tab), Lactose (180 mg/tab) Magnesium Stearate, Microcrystalline Cellulose. Lactose is a sugar from milk. Dextrose is a simple sugar (glucose). If you do not count lactose as a simple sugar then you only have 0.003 g of sugar per tablet. If you count lactose as sugar than you have 0.18 g of sugar per tablet (a minute amount).
SnoreStop FastTabs is prepared in accordance with the Homeopathic Pharmacopoeia of the United States (HPUS). All ingredients are GRAS (generally recognized as safe) and are part of an official collection of accepted drug ingredients by the Federal Food, Drug and Cosmetic Act and are manufactured in FDA regulated laboratories in the USA. Each ingredient derives from plant, mineral or protein sources and is present only in harmless microscopic amounts in each dose. None of our ingredients contain any measurable amount of alkaloids, ephedrine or pseudo - ephedrine.
Dilution Scale: 6X is equal to one part per million.
Claims follow requirement in CPG 400.400 and GLP of Sections 502 & 503 of the FDCA (“The Act”) and Part 201 Title 21 of the CFR to include Principle Display Panel of OTC Drug Labeling under 21 CFR 201.62. Uses and claims are taken from various authorized Materia Medica for references. They have not been reviewed by the Federal Drug Administration.
Reviews
Reviews
Couldn't load pickup availability
Share